Plant Lab
Objective/Purpose : We are looking for plants in our home ecosystem that contain the genetic makeup to create medicines with. This would give us insight on how making real medicine works.
Background: We get the properties used in medicines by tapping into the plants natural chemicals and using it to our advantage inside our own bodies.
Materials List-
Procedure-
water testing paper (w/ plant) methanol testing paper (w/ plant) ampicillin paper water paper
1 Day no no yes no
2 Days no some yes no
Here are my teams results:
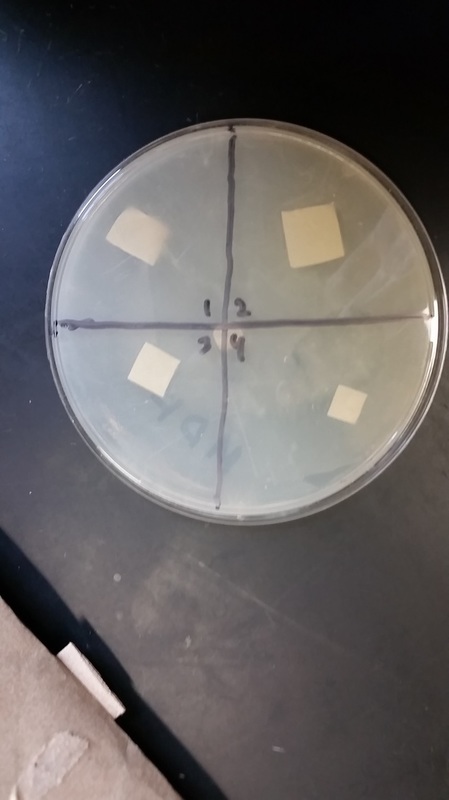
FIRST DAY
Meth Plate: Q 4 has most Bacteria Growth, all other q’s have minimal but some bacteria growth. Rings are around all samples in the agar plate.
Water Plate: Clearance has developed around the positive control and the negative control is showing no clearance. Sample one is also showing no clearance. Samples 2 and 3 are just barely showing clearance
SECOND DAY
Meth Plate: Q4 has most clearance, all others have less but some.
Water Plate: Negative control showing no clearance, Positive control showing lots of clearance. Sample one is showing very little, samples 2 and 3 are showing moderate clearance.
THIRD DAY
Meth Plate: Q4 has most clearance (ampicillin), all others are relatively similar
Water Plate: Quadrants 1 and 2 have a more developed lawn. Sample one still has minimal clearance, sample 2 an hard 3 have a lot of clearance. The negative and positives and controls are showing no clearance and lots of clearance, respectively.
Analysis/Conclusion:
We had a clearing around the positive control. I think that our bacteria field is complete but not for sure. If we didn’t have a complete lawn of bacteria, this disc probably did not absorb enough of the ampicillin . The bacteria grew around the negative control disks. There was a very little clearance around our methanol plant extracts, and none that we could tell around the water plant extracts. Water is not expected to have a negative control disk or antimicrobial activity because water doesn’t kill bacteria, which means there is not any clearance around the paper Something that could have affected our results was a chance of contamination because of the bacteria on the desks or on our hands. Our plant could have antimicrobial activity, but we don’t know because we haven’t tested it. Our results tell us that, lemon leaves won't have any antimicrobial activity, because there was no or very little clearance around the extracts.
water testing paper (w/ plant) methanol testing paper (w/ plant) ampicillin paper water paper
1 Day no no yes no
2 Days no some yes no
Background: We get the properties used in medicines by tapping into the plants natural chemicals and using it to our advantage inside our own bodies.
Materials List-
- Balance, weigh boat, lab scoops
- LB broth base
- Media bottles, 250 mL
- Sterilizer/ autoclaves
- Water bath, 37*C, shaking
- Sterile LB agar
- Laminar flow hood and disinfectant
- Plastic safety glasses
- Bunsen burner and gas lighter
- Inoculating loop, Ni/Cr wire
- Petri dishes, 60x15mm, sterile
- E. coli JM109 (stock plate)
- Plant specimen
- Mortar and pestle
- Pipet, 10 mL and pump
- Plastic funnels, short-stemmed
- Filter paper disks, 5mm diameter
- 100 mL beakers
- Syringe, 10 mL and filter, 0.2 micrometers
- Reaction tubes and rack, 1.7 mL
- Methanol, absolute
- Pipet, 1 mL and pump
- Dry block heater/heat block
- Forceps, fine-tipped
- Ampicillin
- Glass spreader
- Incubator oven,
- 37*C
- Lemon leaves
Procedure-
- Grind up 2 grams of plant tissue from leaves or bark with your mortar and pestle with 10 mL of deionized water. Rest for 3 minutes. Then filter the sample through the 11 cm filter paper funnel. With a syringe filter, sterilize the filtered sample extract. Collect 1 mL of the extract into a 1.7 mL microbe, and be sure to label it.
- Do step 4 again, but replaced the deionized water with methanol. After you extract the methanol, place the 1.7 mL tube with the extract in a 65*C heat block with caps open for 24+ hours to evaporate the methanol.
- Sterilization: Attach pre-filter to syringe and rinse with water. Take to Laminar Hood (plant extract syringe/prefilter, pipet). Label microfuge tube. (initials, W or M). Attach sterile filter to pre-filter. Load 1.7 ml of extract into syringe using pipet. Depress plunger- at least 1 ml. Snap on cap without touching inside.
- For the rest of your samples, do steps 4 and 5 again, making sure to label all samples. There should be 2 tubes in total.
- Evaporate methanol from methanol extract by placing tube, with cap open, on a 65 C heat block overnight.
- Reconstitute methanol extract with 1.0 ml sterile deionized water
- With sterilized forceps that have been flamed in alcohol, drop 3 filter paper disks into every tube of filtered extract.
- Make the negative control disks: three each of only the methanol and only the distilled water.
- Make 2 positive control disks of the ampicillin solution.
- Let the disks soak up enough extract to be saturated. This may have to happen overnight.
- Close all tubes, and store the samples at 4*C until time to use.
- With a sterile pipet, transfer 1 mL of the prepared E. coli broth to the middle of the Petri dish. Then get a glass spreader, sterilize it using alcohol and flame, and spread the broth evenly across the dish. Cover and allow the culture to soak into the agar for at least 15 minutes.
- With sterile forceps (alcohol and flame), place one disk into the middle of each quadrant (which you should have drawn in). Blot out the extra liquid on the disks before you place them on the Petri dish. Keep the methanol-extracted samples on one side and the water on the other.
- Put one of the negative control disks (with methanol or distilled water) in the marked area. Do the same with the positive ampicillin soaked one.
- Let the Petri dish rest for at least a day.
- Examine each quadrant and the controls for areas of inhibition. Photograph or draw your results.
- Depending on your results, make qualitative and quantitative observations and record.
water testing paper (w/ plant) methanol testing paper (w/ plant) ampicillin paper water paper
1 Day no no yes no
2 Days no some yes no
Here are my teams results:
FIRST DAY
Meth Plate: Q 4 has most Bacteria Growth, all other q’s have minimal but some bacteria growth. Rings are around all samples in the agar plate.
Water Plate: Clearance has developed around the positive control and the negative control is showing no clearance. Sample one is also showing no clearance. Samples 2 and 3 are just barely showing clearance
SECOND DAY
Meth Plate: Q4 has most clearance, all others have less but some.
Water Plate: Negative control showing no clearance, Positive control showing lots of clearance. Sample one is showing very little, samples 2 and 3 are showing moderate clearance.
THIRD DAY
Meth Plate: Q4 has most clearance (ampicillin), all others are relatively similar
Water Plate: Quadrants 1 and 2 have a more developed lawn. Sample one still has minimal clearance, sample 2 an hard 3 have a lot of clearance. The negative and positives and controls are showing no clearance and lots of clearance, respectively.
Analysis/Conclusion:
We had a clearing around the positive control. I think that our bacteria field is complete but not for sure. If we didn’t have a complete lawn of bacteria, this disc probably did not absorb enough of the ampicillin . The bacteria grew around the negative control disks. There was a very little clearance around our methanol plant extracts, and none that we could tell around the water plant extracts. Water is not expected to have a negative control disk or antimicrobial activity because water doesn’t kill bacteria, which means there is not any clearance around the paper Something that could have affected our results was a chance of contamination because of the bacteria on the desks or on our hands. Our plant could have antimicrobial activity, but we don’t know because we haven’t tested it. Our results tell us that, lemon leaves won't have any antimicrobial activity, because there was no or very little clearance around the extracts.
water testing paper (w/ plant) methanol testing paper (w/ plant) ampicillin paper water paper
1 Day no no yes no
2 Days no some yes no